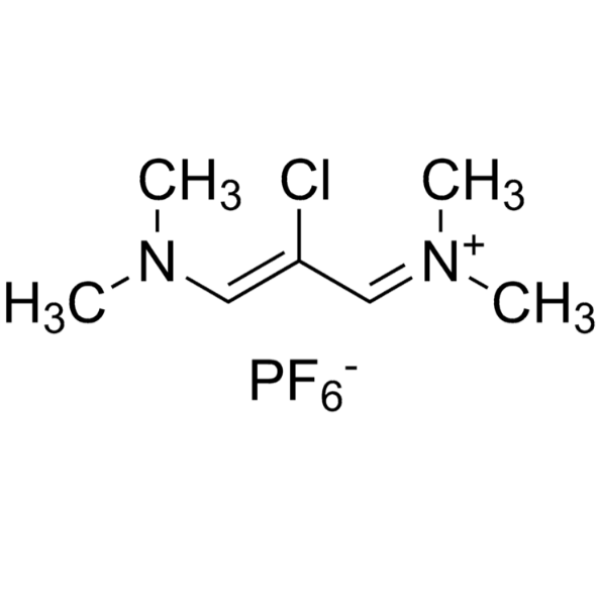
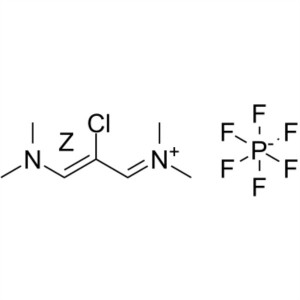
Etoricoxib Intermediate CAS 249561-98-6 ; 291756-76-8 Purity ≥99.0% (HPLC) Factory
Shanghai Ruifu Chemical Co., Ltd. is the leading manufacturer of 2-Chloro-1,3-Bis(dimethylamino)trimethinium Hexafluorophosphate (CAS: 249561-98-6; 291756-76-8) with high quality, intermediate of Etoricoxib (CAS: 202409-33-4). Etoricoxib is used in the treatment of osteoarthritis (OA), acute gouty arthritis, and other diseases.
Ruifu Chemical has been supplying pharmaceutical intermediates and APIs more than 15 years. Ruifu Chemical can provide worldwide delivery, competitive price, excellent service, small and bulk quantities available. Purchase Etoricoxib intermediates, Please contact: alvin@ruifuchem.com
| Chemical Name | 2-Chloro-1,3-Bis(dimethylamino)trimethinium Hexafluorophosphate |
| Synonyms | (Z)-2-Chloro-1,3-Bis(dimethylamino)trimethinium Hexafluorophosphate; (Z)-N-[2-Chloro-3-(Dimethylamino)allylidene]-N-Methylmethanaminium Hexafluorophosphate; N-(2-Chloro-3-(Dimethylamino)allylidene)-N-Methylmethanaminium Hexafluorophosphate(V) |
| Impurity | Etoricoxib Related Compound C |
| Stock Status | In Stock, Commercial Production |
| CAS Number | 249561-98-6 |
| Alternative CAS Number | 291756-76-8 |
| Molecular Formula | C7H14ClN2·F6P |
| Molecular Weight | 306.62 g/mol |
| Melting Point | 125.0~126.0℃ |
| Store Under Inert Gas | Store Under Inert Gas (Nitrogen or Argon) at 2~8℃ |
| COA & MSDS | Available |
| Origin of Product | Shanghai, China |
| Product Categories |
Pharmaceutical Intermediates |
| Brand | Ruifu Chemical |
| Items | Specifications | Results |
| Appearance | Light-Yellow to Brown Powder | Light-Yellow Powder |
| Chloride | ≤0.05% | <0.05% |
| Water by Karl Fischer | ≤0.50% | 0.11% |
| Purity / Analysis Method | ≥99.0% (HPLC) | 99.9% |
| Infrared Spectrum | Conforms to Structure | Conforms |
| NMR Spectrum | Conforms to Structure | Conforms |
| Conclusion | The product has been tested and complies with the given specifications | |
| Application | Intermediate of Etoricoxib (CAS: 202409-33-4) | |
Package: Bottle, Aluminium foil bag, 25kg/Cardboard Drum, or according to customer's requirement.
Storage Condition: Keep the container tightly closed and store in a cool, dry (2~8℃), well-ventilated warehouse away from incompatible substances. Keep away from sunshine; avoid fire and heat sources; avoid moisture.
Shipping: Deliver to worldwide by air, by FedEx / DHL Express. Provide fast and reliable delivery.
How to Purchase? Please contact Dr. Alvin Huang: sales@ruifuchem.com or alvin@ruifuchem.com
15 Years Experience? We have more than 15 years of experience in the manufacture and export of a wide range of high quality pharmaceutical intermediates or fine chemicals.
Main Markets? Sell to domestic market, North America, Europe, India, Korea, Japanese, Australia, etc.
Advantages? Superior quality, affordable price, professional services and technical support, fast delivery.
Quality Assurance? Strict quality control system. Professional equipment for analysis include NMR, LC-MS, GC, HPLC, ICP-MS, UV, IR, OR, K.F, ROI, LOD, MP, Clarity, Solubility, Microbial limit test, etc.
Samples? Most products provide free samples for quality evaluation, shipping cost should be paid by customers.
Factory Audit? Factory audit welcome. Please make an appointment in advance.
MOQ? No MOQ. Small order is acceptable.
Delivery Time? If within stock, three days delivery guaranteed.
Transportation? By Express (FedEx, DHL), by Air, by Sea.
Documents? After sales service: COA, MOA, ROS, MSDS, etc. can be provided.
Custom Synthesis? Can provide custom synthesis services to best fit your research needs.
Payment Terms? Proforma invoice will be sent first after confirmation of order, enclosed our bank information. Payment by T/T (Telex Transfer), PayPal, Western Union, etc.
Hazard Symbols Xi - Irritant
Risk Codes 36/37/38 - Irritating to eyes, respiratory system and skin.
Safety Description
26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice.
WGK Germany 3
2-Chloro-1,3-Bis(dimethylamino)trimethinium Hexafluorophosphate (CAS: 291756-76-8), intermediate of Etoricoxib (CAS: 202409-33-4).
Etoricoxib is a kind of highly selective cyclooxygenase-2 (COX-2) inhibitors developed by the Merck company. Etoricoxib has a unique chemical structure that is methylsulfonyl group. The introduction of this group can not only increase the selectivity for COX-2 drugs, but also does not produce sulfa drugs and cross-allergic reactions.
Etoricoxib was first approved for entering into market in 2002 in the UK, followed by the marketing countries and regions including the European Union, Asia, Australia and Latin America. Until the end of 2013, it has been approved for marketing in 97 countries for being widely used in treatment of osteoarthritis (OA), rheumatoid arthritis, ankylosing spondylitis, chronic low back pain, acute gouty arthritis, primary dysmenorrhea and postoperative pain, and other diseases.
Etoricoxib has also entered into market in Taiwan and Hong Kong of China. It had entered into market in Chinese mainland in 2008 with the approved indications being acute gouty arthritis and OA and another indication being primary dysmenorrhea in the second half year of 2014.
-
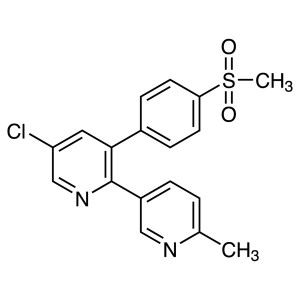
Etoricoxib CAS 202409-33-4 Purity ≥99.0% (HPLC)...
-
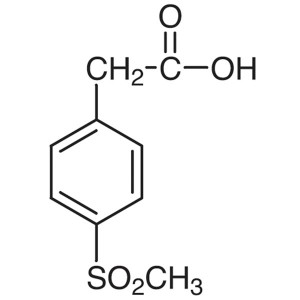
4-(Methylsulfonyl)phenylacetic Acid CAS 90536-6...
-
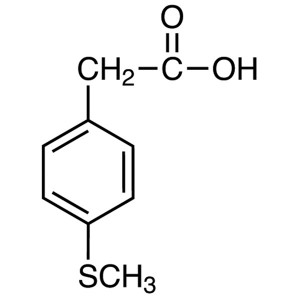
4-(Methylthio)phenylacetic Acid CAS 16188-55-9 ...
-
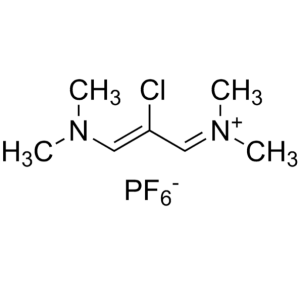
Etoricoxib Intermediate CAS 249561-98-6 ; 29175...
-
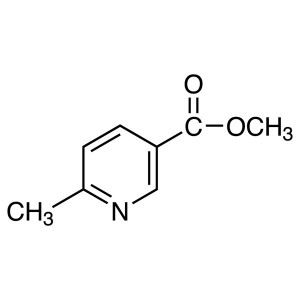
Methyl 6-Methylnicotinate CAS 5470-70-2 Purity ...
-
![1-(6-Methylpyridin-3-yl)-2-[4-(Methylthio)phenyl]ethanone CAS 221615-72-1 Etoricoxib Intermediate Purity >98.0% (HPLC)](https://www.ruifuchem.com/uploads/1-6-Methylpyridin-3-yl-2-4-Methylthiophenylethanone-CAS-1949-89-9-Etoricoxib-Intermediate-Factory-Shanghai-Ruifu-Chemical-Co.-Ltd.-www.ruifuchem.com_-300x300.jpg)
1-(6-Methylpyridin-3-yl)-2-[4-(Methylthio)pheny...
-
![1-(6-Methylpyridin-3-yl)-2-[4-(Methylsulfonyl)phenyl]ethanone CAS 221615-75-4 Etoricoxib Intermediate Purity >98.0% (HPLC)](https://www.ruifuchem.com/uploads/1-6-Methylpyridin-3-yl-2-4-Methylsulfonylphenylethanone-CAS-221615-75-4-Etoricoxib-Intermediate-Factory-Shanghai-Ruifu-Chemical-Co.-Ltd.-www.ruifuchem.com_-300x300.jpg)
1-(6-Methylpyridin-3-yl)-2-[4-(Methylsulfonyl)p...